|
11/25/2023 0 Comments Cathode and anode chargeIt would also offer 30 percent more range. A solid-state battery would be able to charge in a little less than half the time an NMC cell demands. According to Blome, the ID.3 with the biggest battery pack has 100 kilograms (220.5 pounds) in anodes alone.īlome also compared solid-state batteries to NMC cells when it relates to charging times and range. The difference in size is remarkable and makes a good example of how much lighter solid-state batteries have the potential to be. The Volkswagen executive presented their structure compared to that of an NMC cell. Anyway, Volkswagen wants to have vehicles with batteries that start charging with an anodeless design: solid-state batteries. The Porsche Taycan and the Audi E-Tron GT already use cells with silicon in their anodes, making them spend about 30 percent less time charging and have 10 percent more range than the vehicles without that solution. You should put the thickest part of your patient thus toward the cathode to take advantage of the heel effect. The primary beam is thus heavier on x-rays on the side toward the cathode & weaker (less photons) on the anode side. Volkswagen’s idea is to add silicon to that recipe, something Oliver Blume said Porsche is pursuing as well. Erase the part of the 360 degree bean then that is taken out by the anode. Most anodes are currently made of synthetic graphite. Regarding the anodes, they have an impact of only 10 percent in range, while charging time depends 100 percent on them, according to Blome. This is the chemistry Volkswagen wants to adopt for volume EVs. Manganese is a cheaper metal than nickel, and it can provide the same range of NMC cells for about 80 percent of the cost, just like LFP. Blome stresses that its advantages are not only in cost but also in cycling stability and safety.Ĭontrary to what most companies say, Blome said Volkswagen would pursue high-manganese cells, not high-nickel batteries. On the other hand, it offers only around 80 percent of the range. SEM images of cross-section of the (a) as coated NMC cathode, (b) calendered NMC cathode, (c) as coated graphite anode and (d) calendered graphite anode electrodes. Chlorine will form at the positive electrode because non-metals form there from negatively charged non-metal ions.Compared to an NMC cell, a lithium iron phosphate battery has about 80 percent of the cost – which is a good thing. Predict the products of electrolysis of molten calcium chloride.Ĭalcium will form at the negative electrode because metals form there from positively charged metal ions. Lead and bromine form during the electrolysis of molten lead bromide 
So lead forms at the negative electrode and bromine forms at the positive electrode. Br - ions lose electrons at the anode and become Br atoms, which pair up to form Br 2 molecules.Pb 2+ ions gain electrons at the cathode and become Pb atoms.the anode and the cathode are marching more quickly but at nearly. 
Molten lead bromide, PbBr 2 (l), is an electrolyte. The data provided in this manuscript shows that although the charge and discharge endpoints march faster when the anode loading is higher (R 4.54), the rate of capacity fade is actually lower since the difference in marching rates of the anode and the cathode is smaller, i.e. anions lose electrons at the positively charged anode.cations gain electrons from the negatively charged cathode.As a result, they form atoms or molecules of elements: When ions reach an electrode, they gain or lose electrons. Ions migrate (move) towards their oppositely charged electrode Products of electrolysis 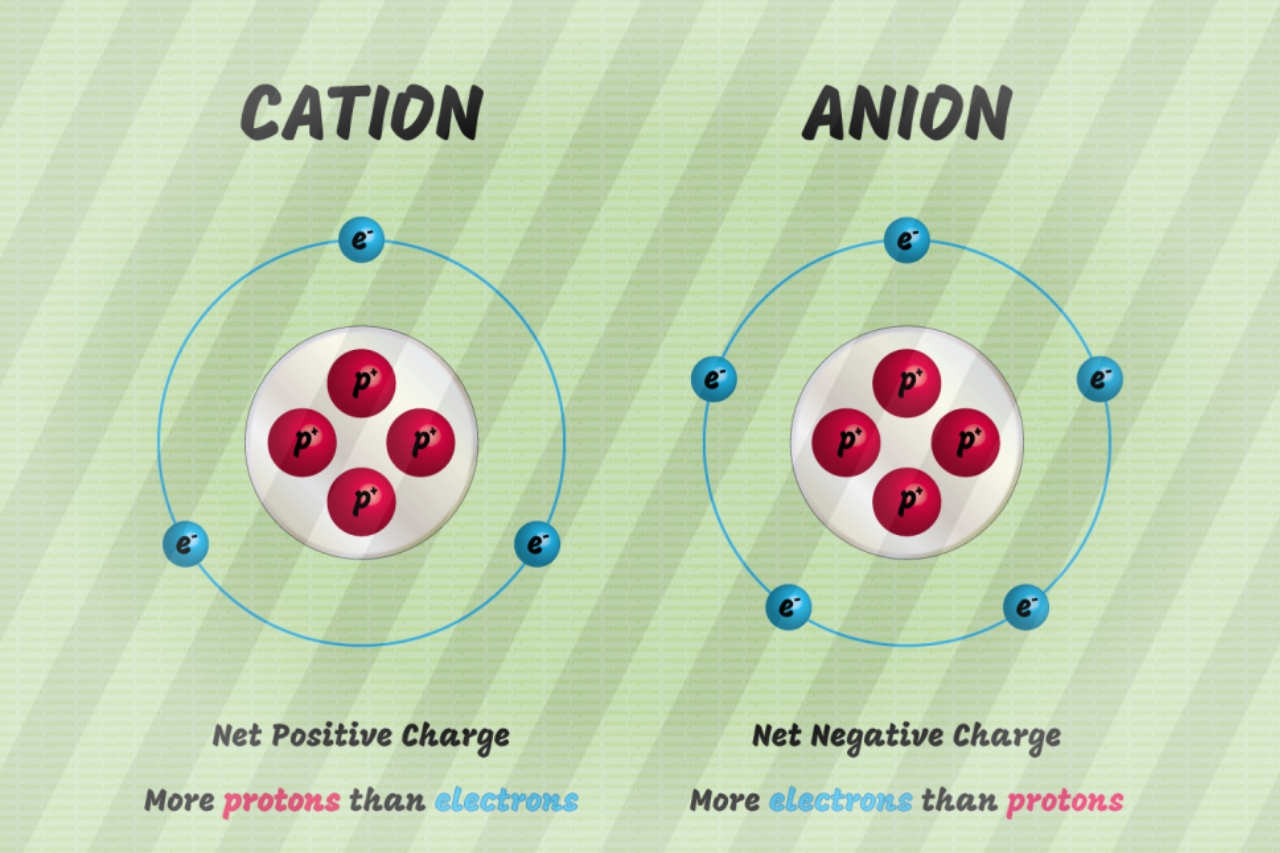
Negatively charged ions are called anions. The positively charged electrode in electrolysis is called the anode. Positively charged ions are called cations. The negatively charged electrode in electrolysis is called the cathode. 
The free ions in electrolytes are attracted to the oppositely-charged electrodes connected to the dc supply. Under these conditions, the ions in electrolytes are free to move within the liquid.Įlectrolysis is a process in which electrical energy, from a direct current (dc) supply, decomposes (breaks down) electrolytes. Electrolytes are ionic compounds that are either:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
